First-order kinetics in a PFR
Environmental engineering
Problem statement
Lets consider 2 plug flow reactors (PFRs) with constant cross-sectional area \(A\) and length \(L\), where a substance decays with a first-order rate constant \(k_1\). Each reactor receives an inflow \(Q\) with a concentration of substance \(C_{in}\), while the outflow equals the inflow. The parameters for each PFR are the following:
- PFR 1: \(Q\) = 5 L/s, \(A\)= 0.2 m\(^2\), \(C_{in}\) = 12 mg/L, \(k_1\) = 0.1 s\(^{-1}\)
- PFR 2: \(Q\) = 5 L/s, \(A\)= 0.4 m\(^2\), \(C_{in}\) = 12 mg/L, \(k_1\) = 0.1 s\(^{-1}\)
- Compute the mean flow velocity in each reactor.
- Plot the concentration in each reactor as a function of position.
- Determine the concentration in the reactor at \(x\) = 0.5 m.
- Plot the accumulated mass in each reactor as a function of position.
Solution
For a PFR with constant area \(A\) and flow \(Q\), the mean flow velocity is equal to \(v = Q/A\). For an ideal PFR we assume perfect radial mixing, but no mixing along the flow direction. Thus, the concentration inside the reactor varies as a function of position. By considering a moving control volume \(dV = Adx\), located at position \(x=vt\), the concentration of a substance \(C\) that decays with a first-order rate constant \(k_1\) can be modeled as follows:
\[ \dfrac{dC}{dt}dV = QC - QC - k_1 C dV \tag{1}\]
Note that the relation between time and position \(x\) of the control volume is given by the mean flow velocity \(v\):
\[ dt = \dfrac{dx}{v} \tag{2}\]
Replacing Equation 2 in Equation 1 and canceling terms yields the following ordinary differential equation (ODE):
\[ \dfrac{dC}{dx} = -\dfrac{k_1}{v} C \tag{3}\]
Which is analogous to a CMBR reactor with first-rate order kinetics. Assuming that at \(x=0\) the concentration of substance in the reactor is \(C_{in}\), then Equation 3 has the following analytical solution:
\[ C(x) = C_{in} e^{-\frac{k_1}{v}x} \tag{4}\]
Note that the concentration of substance inside the reactor does not depend on the initial concentration in the reactor, since it gets completely washed by the inflow. In addition, the equation allows to compute the concentration inside the reactor regardless of its length \(L\).
Lets implement Equation 4 numerically through the following function:
Note that the function conc_pfr() receives the flow velocity v, the inflow concentration Cin, the first-order decay rate constant k1, and the position array x.
Flow velocity
To compute the flow velocity we simply need to divide the flow \(Q\) by the cross-sectional area \(A\):
PFR 1: A = 0.2 m^2; v = 2.50 cm/s
PFR 2: A = 0.4 m^2; v = 1.25 cm/sThus, for a PFR with a given flow \(Q\), the mean flow velocity \(v\) is inversely proportional to the cross-sectional area \(A\).
Plot of concentration
fig, ax = plt.subplots(figsize=(3,3)) # figure size
x = linspace(0, 1) # pos in m
ls = ['-', '--'] # line styles
for i, v in enumerate(vel):
Cx = conc_pfr(v, Cin, k1, x)
ax.plot(x, Cx, color='k', ls=ls[i],
label="$A$ = %s m$^2$" % A[i])
ax.axvline(0.5, color='k', ls=':')
ax.set_xlabel("position [m]")
ax.set_ylabel("concentration [mg/L]")
ax.set_xlim(x.min(), x.max())
ax.legend(edgecolor='k', fontsize=9)
ax.grid()
plt.show()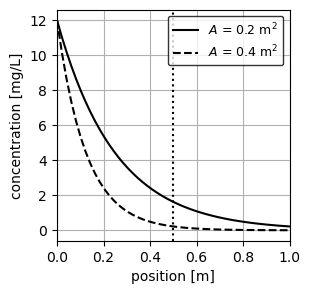
As expected from Equation 4, the concentration of each PFR decays exponentially as a function of position, with the reactor with greater area decaying faster owing to a greater residence time (\(\tau_x=Ax/Q\)). We additionally plot a vertical line at \(x\) = 0.5 m to visually inspect the concentration at this position in the reactor.
Concentration at \(x\) = 0.5 m
We can use the function conc_pfr() to determine the exact concentration of both PFR at \(x\) =0.5 m.
Accumulated mass of substance in the PFR
To compute the accumulated mass of substance in the reactor we simply need to integrate Equation 4 with respect to a given volume:
\[ M(x) = \int_{0}^{V} C(x)dV = \int_{0}^{x} C_{in}e^{-\frac{k_1}{v}x} A dx \tag{5}\]
\[ M(x) = \dfrac{C_{in}Q}{k_1}\left(1 - e^{-\frac{k_1}{v}x}\right) \tag{6}\]
We implement Equation 6 to plot the mass of substance in the PFR as a function of position:
Now lets plot the accumulated mass of substance in each PFR:
fig, ax = plt.subplots(figsize=(3,3)) # figure size
x = linspace(0, 1) # pos in m
ls = ['-', '--'] # line styles
for i, v in enumerate(vel):
Mx = mass_pfr(v, Cin, Q, k1, x)
ax.plot(x, Mx/1e3, color='k', ls=ls[i],
label="$A$ = %s m$^2$" % A[i])
ax.axvline(0.5, color='k', ls=':')
ax.set_xlabel("position [m]")
ax.set_ylabel("mass [g]")
ax.set_xlim(x.min(), x.max())
ax.legend(edgecolor='k', fontsize=9)
ax.grid()
plt.show()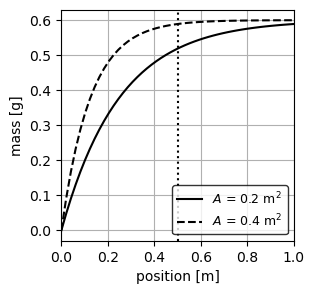
Follow up questions
- Verify that when \(L=v/k_1\) the outflow concentration of the PFR is \(C_{in}/e\).
- Verify that the maximum accumulated mass of substance in a PFR is \(C_{in}Q/k_1\), regardless of reactor geometry.
- Plot the adimensional relation between \(C/C_{in}\) as a function of \(k_1x/v\). What is the required value of \(k_1x/v\) to decrease the inflow concentration in 90%?